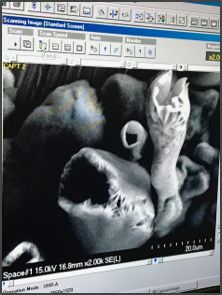
“Who knew rust could be so beautiful!” remarked Kai, as the image emerged on the screen connected to the Scanning Electron Microscope (SEM). On invitation, Kai Turner and Alec Lewis, recent high school graduates from iLEAD’s Santa Clarita Valley International Charter School (SCVi), in Castaic, CA, accompanied by Kathleen Fredette, Director of STEAM Initiatives for iLEAD Schools, traveled to NASA Glenn Research Center to collaborate with Material Chemist, Bryan Harder.
“Science is not simply about figuring out or discovering a fact that nobody else ever considered. It’s about standing on the shoulders of giants, adding another piece to the story, which is our collective understanding of how things work, what stuff is like, where we came from and where we’re going,” said Harder. This is just one of the lessons learned during the two day visit, as Harder guided the team through an analysis of their iron samples, an experiment, which had recently been activated and conducted on International Space Station.

As part of Student Spaceflight Experiments Program, SSEP, Turner, Lewis (along with team member, Dustin Fields) wrote an experiment proposal that was carried out by astronaut, Jack Fischer. Simultaneously and back on Earth, the SCVi team activated their identical ground-based experiment so both samples could be analyzed and compared to its space-based counterpart.
Through Harder’s guidance, the learners strategized the progression of instruments they would use to examine the simple bars of iron, only two inches long. The instruments included the SEM, which reveals the surfaces using an electron beam; a x-ray diffractometer, providing insight into the kind and type of crystalline formation of the oxides; and an optical profilometer, which uses light to determine surface roughness and morphology. The team’s initial analysis was reported, with the final analysis to be published this year.
Alec, who now attends Florida Polytechnic University, attributes his acceptance and scholarship to his involvement in this project. “I think I learned more about authentic science in the two days we spent with Bryan than we did during all of high school.”

This is the reason it’s imperative to connect industry experts with young people rather than simply reading text or watching science videos. It’s about getting in there, doing science or any craft for that matter; not waiting for your learners to arrive at a university and then begin to do the real deal stuff. It’s about inspiration, mentoring and igniting intellectual sparks. Education is at its best when it provides platforms for young people to be immersed in authentic projects, giving them a reason to pursue their education and find delight in the unique gifts they bring to their world.
The Student Spaceflight Experiments Program (SSEP) is a program of the National Center for Earth and Space Science Education (NCESSE) in the U.S. It is enabled through a strategic partnership with DreamUp PBC and NanoRacks LLC, which are working with NASA under a Space Act Agreement as part of the utilization of the International Space Station as a National Laboratory.
